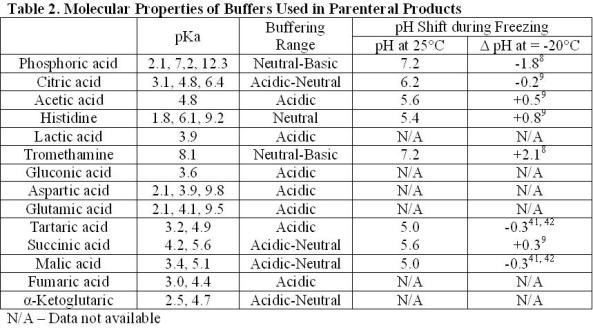
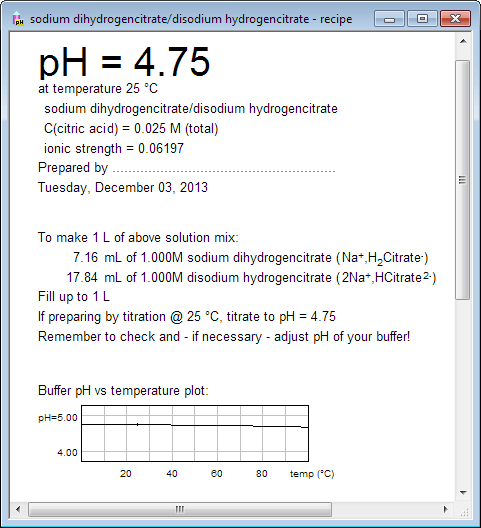
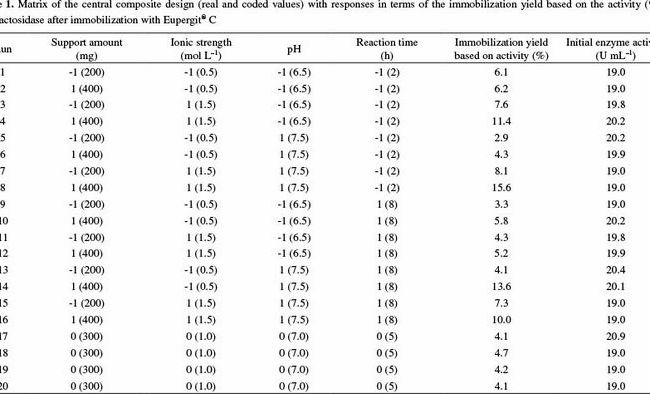
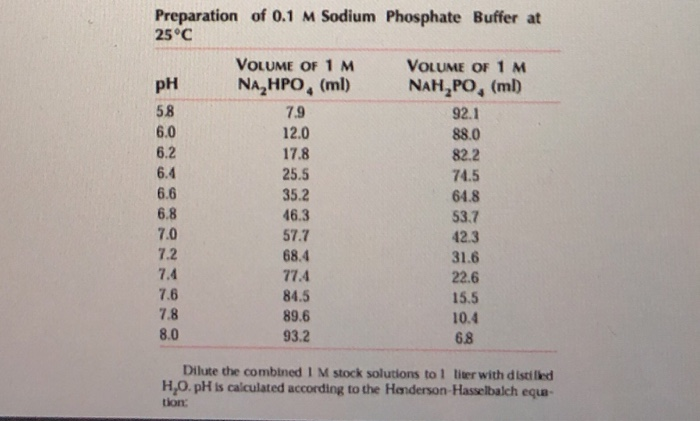
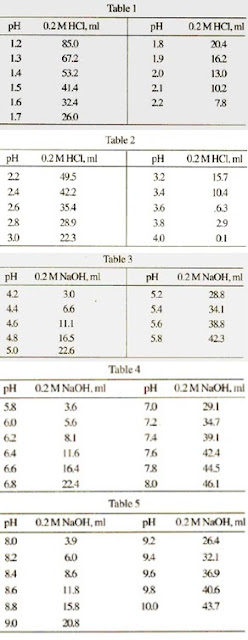
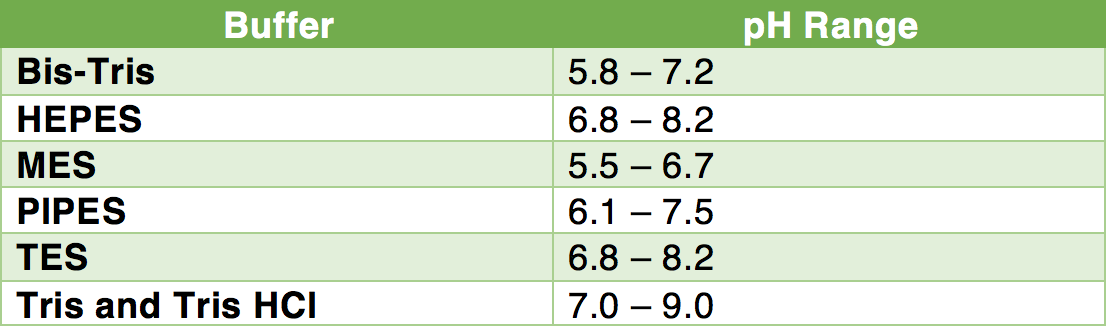
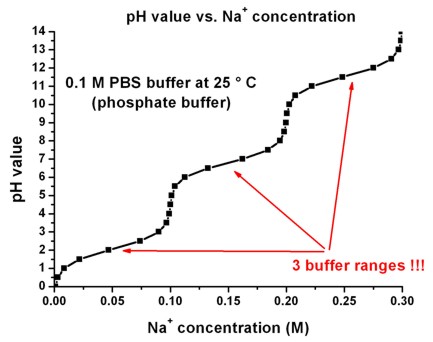
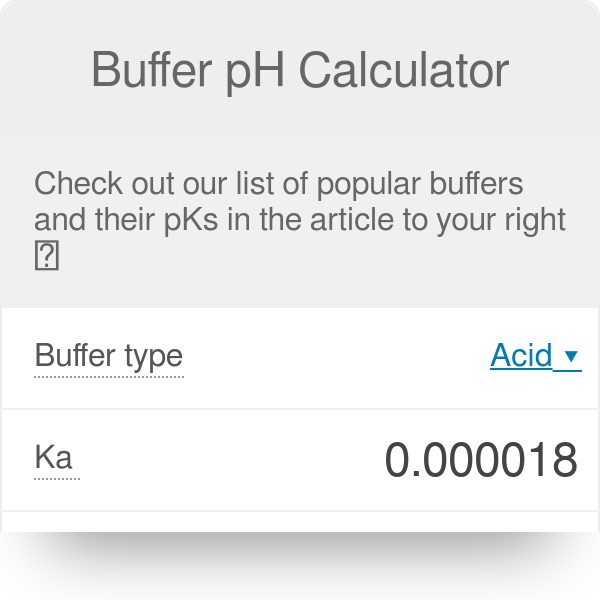
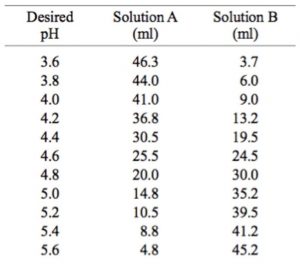
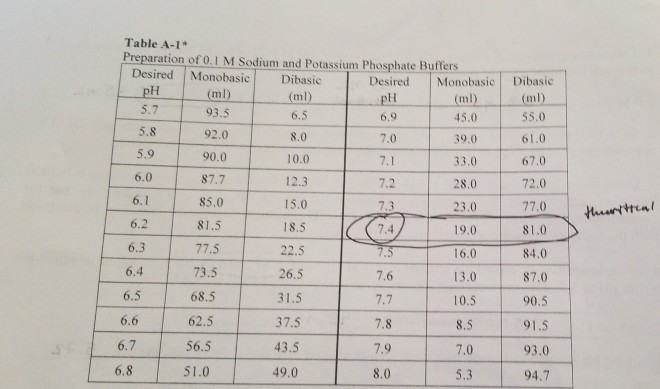
pH calculations and more in fundamentals of pharmaceutics. : Prepare 1 liter phosphate buffer solution at pH = 7.4 and with a buffer capacity of 0.1
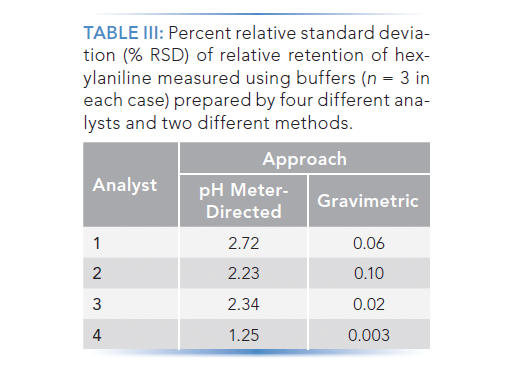
Mobile Phase Buffers in Liquid Chromatography (LC): Effect of Buffer Preparation Method on Retention Repeatability
The pKa of the phosphate buffer system is 7.2. Using the Henderson Hasselbach equation, what is the final pH of the environment after adding 25 mL of 0.01 M HCl to 1